|
6/14/2023 0 Comments Workdone in thermodynamics
We will assume the expansion process is quasi-equilibrium, and the piston moves up an infinitesimal distance d. is to make it possible to work through the text in a variety of orders and at. Upon receiving heat, the gas will tend to expand, pushing the piston up. shift in emphasis away from classical thermodynamics by combining several. The gas in the cylinder exerts an upward force, F=PA, where P is the gas pressure, and A is the cross-sectional area of the piston. Part of such surroundings-defined work can have a mechanism just as for system-defined thermodynamic work done by the system, while the rest of such. If the volume of the gas changes from V1 to V2, then the total work done by the gas depends on how exactly the pressure varies during the expansion process. 
Let us consider a piston-cylinder device, as illustrated in Figure 4.3.2. In thermodynamics, the work done is given by wdVPext For a system undergoing a particular process, the work done is, wdV(VbRTV2a) This equation. 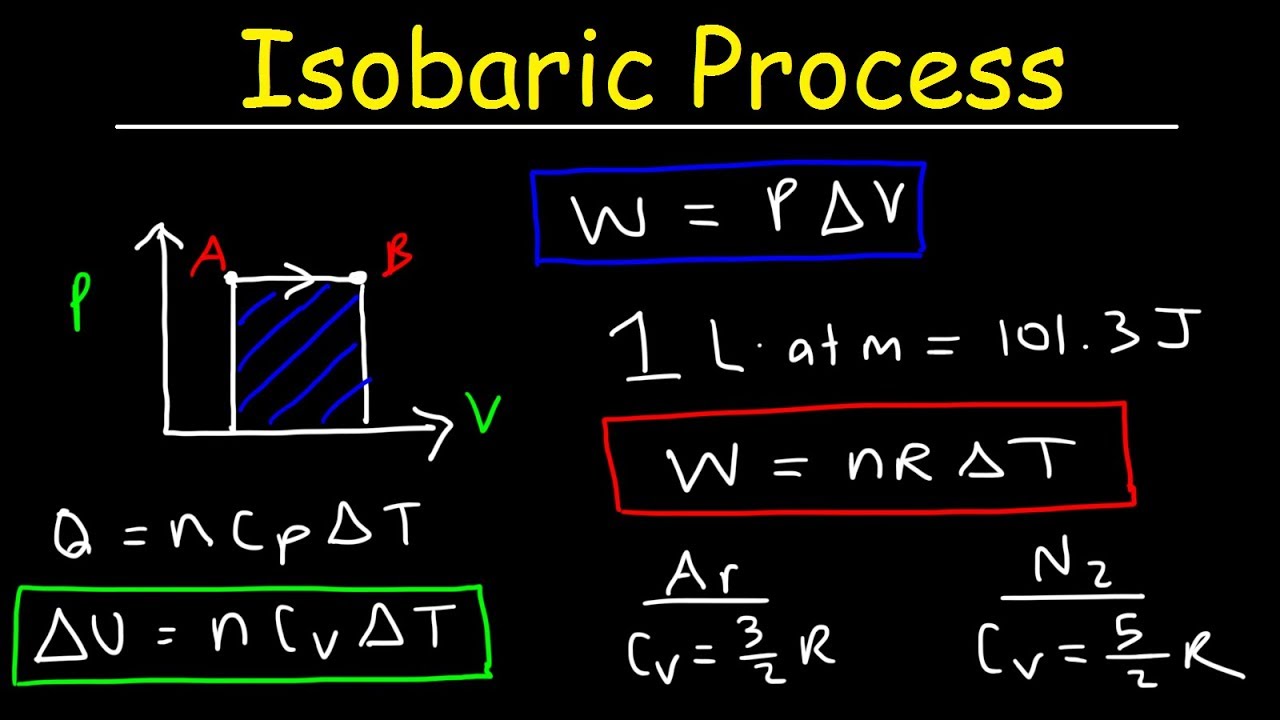
Work associated with the expansion and compression of a gas is commonly called boundary work because it is done at the boundary between a system and its surroundings. Figure 4.3.1 Work done due to a force acting on a block over a distance 4.3.1 Boundary work
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
